Chemical Bond: It is the force which keeps atoms together in a molecule. The causes of formation of a chemical bond are:
Tendency of atoms to complete their octets or duplets by rearrangement of their valence electrons (octed theory).
The system acquires minimum energy when atoms are at some equilibrium distance where attractive forces dominate over repulsive forces.
Types of Chemical Bonds:
Ionic bond
Covalent bond
Metallic bond
Co-ordinate bond
Ionic Bond:
a) Favourable conditions to form Ionic Bond:
Formed between electro positive element (group 1,2,13) and electro negative element (group 15,16,17)
 E.N >=2
E.N >=2Lower I.E of one atom and high E.A of second atom
Higher lattice energy
Larger cations, smaller anions
b) Properties: Crystaline, stronger force of attraction, thermally stable, low volatality, high density, high melting point and boiling point, highly soluble in polar solvent, good conductor of electricity in molten or solution state, nondirectional bond.
Covalent Bond:
a) Favourable conditions to form Covalent Bond:
Formed between two electronegative elements (group 14,15,16,17)
 E.N < 1.9
E.N < 1.9Small cations, larger anions (Fajan’s Rule)
High charges on cation and anion (Fajan’s Rule)
Covalent bond is formed by sharing of electrons by two atoms to complete their octet or duplet
b) Properties:
Compounds containing covalent bonds under normal condition of pressure and temperature exist as gases or liquids of low boiling point due to weak Vander Wall’s force
Relatively low melting point and boiling point, generally non-conductor, and soluble in non-polar solvent
As the bond is rigid and directional, compounds with covalent bond show isomerism
Covalency of the atom is equal to the number of covalent bonds formed by the atom
Bond Length: It is the average distance between the nuclei of two bonded atoms. It depends upon:
Size of the atom: Bond length increases with increase in the size of atom (HI > HBr > HCl > HF)
Multiplicity of bond: Bond length decreases with multiplicity of bond (C-C > C=C )
Type of hybridisation: More s-character, shorter is the bond length, greater is the acidity with comparable compound
Bond Energy: It is the energy required to break one mole of bonds of a particular type of substance in gaseous state.
Bond Angle: It is the internal angle between the orbitals containing electron pairs in the valence shell of the central atom in a molecule:
More the lone pairs on the central atom, smaller is the bond angle due to bp repulsion
More electronegative the central atom, more is the bond angle
More electronegative the surrounding atoms, lesser is the bond angle
Valence Bond Theory (VBT): Proposed by Heitler and London and extended by Pauling and State. The postulates are:
Overlapping of atomic orbitals of valence shells of two atoms leads to the formation of a covalent bond
Half filled orbital and opposite spin electrons are used
 – bond is formed by
head on overlapping,
– bond is formed by
head on overlapping,  – bond is formed by
lateral overlapping
– bond is formed by
lateral overlappingGreater the overlapping, stronger is the bond
 – bond is stronger
than
– bond is stronger
than  – bond and
– bond and  – bond is directional while
– bond is directional while  – bond
is non-directional
– bond
is non-directionalThe directon of the bond is the same as the direction of overlapping of orbitals
The strength of
 – bond follows the order s -s < s –
p < p – p
– bond follows the order s -s < s –
p < p – pPaired electrons are shifted to higher energy levels while forming a bond
VBT cannot explain paramagnetic behaviour of O2
Hybridization:
Number of hybrid orbitals = number of atomic orbitals intermixed
Hybrid orbitals form
 -bond on overlapping
-bond on overlappingIt does not take place in and isolated atom. It occurs only during bond formation
Hybrid orbitals tend to remain far apart therefore repulsion order is lp-lp>lp-bp>bp-bp
Total number of hybrid orbitals of central atom = Number of its
 – bond pairs + Number of lone pair of
electrons around the central atom
– bond pairs + Number of lone pair of
electrons around the central atomDifferent types of hybridization are sp, sp2 , sp3 , sp3d ,sp3 d2 ,sp3 d3
Molecular Orbital Theory (MOT): The theory was developed by Hund & Mulliken. The basic postulates are:
All atomic orbitals are mixed up to form molecular orbitals
Total M.O = Total A.O
Two A.O. combine to form tow M.Os. One is anti bonding M.O. and the other is bonding M.O.
The filling up of electrons must follow Aufbau’s principle., Pauli exclusion principle and Hund’s rule.
Bond order = (Nb – Na )/2 where Nb = Number of electrons in bonding M.O and Nb = Number of electrons in antibonding M.O
Bond order
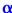 Bond energy
Bond energy  1/Bond length
1/Bond length
Dipole Moment:
Common in covalent compounds when formed between two dissimilar atoms
Used to predict the extent of polarity in a molecule
A vector quantity so follows vector addition or subtraction rule in predicting net dipole moment of a molecule
Can predict the shape of molecules, H2 O – bent, BeF2 – linear
Metallic Bond:
a) Favourable conditions to form Metallic Bond:
Formed between Electro positive ions packed in one of the 3 arrangements (1.CCP(or FCC), 2.HCP, 3 BCC)
b) Properties:
Negatively charged electrons hold the ion together
Highly conducting becauls the mobility of these electrons through the lattice
High m.p and b.p
If soluble, then soluble only in polar solvents of high dielectric constant
Co-ordinate Bond:
a) Favourable conditions to form Metallic Bond:
One of the group or or atom must have a lone pair of electrons whereas the other must have the incomplete octet or duplet.
Also known as dative bond
b) Properties:
Intermediate between ionic and covalent compounds
Sparingly soluble in water, not forming ions, largely soluble in non-polar solvents
Melting and boiling points are higher than purely covalent compounds but less than purely ionic compounds
Stable as covalent compounds, addition compounds are very stable
Non conductor
Bond is rigid and directional
High value of dielectric constant
Other Bonds:
a) H-bonding: When H is attached to N, O, F
It’s strength is about one tenth of a covalent bond
Small size and high electronegativity of atoms forms a strong H-bond
They are of twor types; intermolecular (between two molecules) and intramolecular (within a molecule)
Used to predict boiling point and density of water, solubility of alcohols and carboxylic acids in water.
b) Dipole-dipole interactions:
These exist between molecules having permanent dipoles e.g. H-Cl, I-F etc
c) Ion-dipole interactions:
Between and ion and a polar molecule eg hydration of ions like Na+ , Mg2+ etc
Smaller the ion, more the dipole moment, stronger will be these interactions
d) Ion-induced dipole interactions:
Between an Ion and a dipole induced by the ion in a nonpolar molecule
e) Dipole-induced dipole interactions:
Between a dipole and and induced dipole in a non polar molecule
f) Dispersion forces:
Between two nonpolar substances. The forces are between an instantaneous dipole and and induced dipole eg in O2 , N2 , He, Ne, Ar etc
Note: Dipole-dipole, dipole-induced dipole and dispersion forces are called Van der Wall’s forces